I’ve been a “road warrior” most of my adult life, so the concept of having a “go bag” prepped and ready to grab is nothing new to me. Go bags generally refer to an emergency kit filled with everything you need for two or three days. In a home, it might be batteries, flashlight, water and canned food, along with any medications you need and a change of clothes or two. For business travelers, the idea is that you have a suitcase packed and if something comes up quickly, you simply grab the suitcase and head out. It’s a nice thing to have if you get a lot of last-minute changes to your schedule.
HQAA Blog
Topics: Employee Training, Renewing Accreditation, HME Accreditation Requirements, Emergencies, Marketing
In the past, we’ve reviewed various top ten lists highlighting the most frequently cited standards and/or the most common deficiencies (see Blog articles from March, 2017 & October, 2020). The new year seems like a good time to revisit that old trope and see how we are progressing as an industry. It turns out, the same array of standards and deficiencies continue to challenge many companies.
It’s funny how the Christmas & Holiday season starts earlier and earlier every year. Traditionally, Thanksgiving Weekend was a kickoff to the holiday season: Black Friday shopping, Christmas Parades, Santa arriving at the shopping mall, and lighting the town’s Christmas tree all occurred that weekend. Now, holiday sales start weeks before Thanksgiving and its pretty common for folks to put their Christmas tree up early so their guests can enjoy it on Thanksgiving. Radios blast Christmas music before the leaves are raked and hauled away. Stores sport decorations and sell wrapping paper and tinsel in October and even September.
Zen & The Art of Hospice Contract Maintenance
There was a great book I read in either high school or college titled “Zen & The Art of Motorcycle Maintenance”. Written by Robert Pirseg, it was probably the first truly profound and deep reading I ever did – and it made a lasting impression on me. (Full disclosure: I read it twice in my life; both times were over 35 years ago and I have not re-read it since). The book told the story of a father and son crossing the country on a motorcycle. The motorcycle would break down or need some work or repair and the father son duo worked through it together.
Topics: Quality Care, Oxygen, Equipment
Helpful Resources: How the HQAA Website Can Be Your BFF
HQAA designed their application and accreditation processes to be user friendly. Achieving accreditation, especially for a new startup organization, can be stressful as a company navigates what can seem bureaucratic and complicated waters in pursuit of their accreditation. But, never fear: HQAA has set up easily accessible resources to smooth those processes and guide your organization.
Topics: Renewing Accreditation, HQAA Accreditation, HME Accreditation Requirements, Avoiding Deficiencies
When I was a kid—maybe 6th or 7th grade—I had a friend that could name all the states in alphabetical order. He also could name each state capital. Half a century later, after a career spent traveling the United States, I think I might be able to list them all if I could start with Maine and work geographically south and west visualizing a map. My friend was a pretty smart individual and is finishing up a stellar career as a cardiothoracic surgeon. I often wonder if he can still list the states like that in between heart valve replacement surgeries and repairing aortic aneurisms. And because that special talent so impressed me back in the day, I think about him and his high-intelligence parlor trick every time I see a map of the United States with the outline of states and the Crayola crayon rainbow of colors for each place.
Topics: DMEPOS, State Regulations
When It Absolutely, Positively Needs to Be There Overnight
In the late 1970’s, FedEx, a multi-national conglomerate company which specialized in global delivery services launched an advertising campaign that became their mission statement and philosophy. “When it absolutely, positively has to be there overnight” appeared in their print ads, their television and radio commercials, and on flyers, and signage in their ubiquitous storefront counters. The statement encapsulated the messaging from FedEx: if you can’t afford a delay, use us. FedEx would describe the tagline as “more than a slogan—it was a promise that demanded trust”. FedEx grew meteorically through the late 1970’s and into the 1980’s. Their brand remains today and they are synonymous with not only global delivery, but e-commerce business solutions.
Topics: Billing, Ventilator Unit, Retail, Delivery, Oxygen, Customer Service, Business Practices, Equipment, DMEPOS
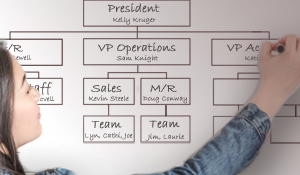
Say Their Names: Identifying Officers & Titled Roles in Your DME
General Colin Powell once famously stated: “Organizational charts and fancy titles count for next to nothing”. Depending on the context or the situation, that may or may not be true. But with all due respect to General Powell, identifying certain titles and positions within your organization and pinning them on an accurate organizational chart is a necessary requirement and the basis of several accreditation standards. The standards require naming the actual persons and titles and reporting mechanism within your organizations.
“Morale” is defined as “the mental and emotional condition of an individual or a group with regard to a task or goal to be accomplished”. Good, positive morale by staff and team members is an essential component of any business plan. Bad, negative morale can ruin a team and literally destroy a business.
Complex or custom rehabilitation technology services are highly specialized services and products designed to assist patients with disabilities to help perform activities of daily living (ADL’s). In the DME world, in terms of DMEPOS, most of the equipment and supplies that fall into this category are used for mobility. Customized wheelchairs with specialized seating surfaces and controls are the products that are most commonly provided.
Topics: HQAA Accreditation, HME Accreditation Requirements, DMEPOS, Complex Rehab