Prior to 2026 (some industry folks would call that era, “the good old days”), when you started a new DME or opened a branch, it was acceptable and even considered best practices to put together MOCK patient files using the paperwork your company planned on using for consideration and evaluation by the surveyor. Because you had to comply with the CMS Supplier Standards, the new location –be it a brand-new startup company or simply a new branch/location for an existing company—was required to be open for business. But because it was --and is-- typically very difficult to find patients willing to self-pay, and even hard to connect with patients who would accept DMEPOS at no charge, the new company was not expected or required to have actual patients.
HQAA Blog
Topics: HME Accreditation Requirements, Showroom, Retail, Marketing, Equipment, DMEPOS
I’ve been a “road warrior” most of my adult life, so the concept of having a “go bag” prepped and ready to grab is nothing new to me. Go bags generally refer to an emergency kit filled with everything you need for two or three days. In a home, it might be batteries, flashlight, water and canned food, along with any medications you need and a change of clothes or two. For business travelers, the idea is that you have a suitcase packed and if something comes up quickly, you simply grab the suitcase and head out. It’s a nice thing to have if you get a lot of last-minute changes to your schedule.
Topics: Employee Training, Renewing Accreditation, HME Accreditation Requirements, Emergencies, Marketing
In the past, we’ve reviewed various top ten lists highlighting the most frequently cited standards and/or the most common deficiencies (see Blog articles from March, 2017 & October, 2020). The new year seems like a good time to revisit that old trope and see how we are progressing as an industry. It turns out, the same array of standards and deficiencies continue to challenge many companies.
Helpful Resources: How the HQAA Website Can Be Your BFF
HQAA designed their application and accreditation processes to be user friendly. Achieving accreditation, especially for a new startup organization, can be stressful as a company navigates what can seem bureaucratic and complicated waters in pursuit of their accreditation. But, never fear: HQAA has set up easily accessible resources to smooth those processes and guide your organization.
Topics: Renewing Accreditation, HQAA Accreditation, HME Accreditation Requirements, Avoiding Deficiencies
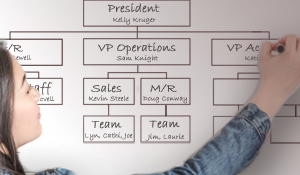
Say Their Names: Identifying Officers & Titled Roles in Your DME
General Colin Powell once famously stated: “Organizational charts and fancy titles count for next to nothing”. Depending on the context or the situation, that may or may not be true. But with all due respect to General Powell, identifying certain titles and positions within your organization and pinning them on an accurate organizational chart is a necessary requirement and the basis of several accreditation standards. The standards require naming the actual persons and titles and reporting mechanism within your organizations.
Complex or custom rehabilitation technology services are highly specialized services and products designed to assist patients with disabilities to help perform activities of daily living (ADL’s). In the DME world, in terms of DMEPOS, most of the equipment and supplies that fall into this category are used for mobility. Customized wheelchairs with specialized seating surfaces and controls are the products that are most commonly provided.
Topics: HQAA Accreditation, HME Accreditation Requirements, DMEPOS, Complex Rehab
One of my funniest memories from surveying was many years ago at a small DME company in the Midwest. The staff was pleasant and accommodating and had been well prepared for the survey. But for some reason (probably related to a co-worker’s retelling of a “bad” survey that she’d gone through), the staff was pretty nervous about the accreditation visit. I pride myself on NOT presenting an intimidating attitude, but the staff at this place thought I was the police, the IRS, and the guy in charge of the Inquisition rolled into one. I tried my best to put the staff at ease. At one point, I asked a customer service representative about how they conveyed information on the patient’s rights and responsibilities to new customers. She fumbled through a pretty good answer. I asked her if she could name one of the responsibilities and she answered that they need to inform the company if they move or if their insurance changes (a good answer). I asked her if she could name a right and she froze up. Finally, she took a deep breath and blurted out: “The right to remain silent”.
Topics: Employee Training, Billing, HME Accreditation Requirements, Clinical Practice Guidelines, Retail, Delivery
DME owners and managers often cite quality improvement (QI) and/or performance improvement (PI) as one of the most difficult concepts to understand and one of the most difficult programs to implement within their businesses. And industry consultants and surveyors find the so-called QI Standards some of the most frequently cited standards for deficiencies and recommendations, and one of the areas in which they spend the most consultative time educating companies.
Topics: Quality Improvement, HME Accreditation Requirements, Process Improvement, Customer Service, Business Practices
I’m not going to name names, but many years ago, as a young surveyor, I was inspecting delivery vehicles at a place (the location will remain unnamed) and observed one of the worst safety hazards I ever saw in my career. A delivery technician who covered a two-state territory had been in the adjacent state where fireworks were legal, and decided to stock up for the
Topics: HME Accreditation Requirements, Avoiding Deficiencies, Delivery, Oxygen, Equipment
Mission & Vision: The First Standard for a Reason
The very first standard in the HQAA Accreditation Standards is ORG 1 MISSION & VISION. The “mission & vision” standard is the first standard for a reason: the mission and vision set the tone for your organization’s corporate culture and define who you are and why you exist. They represent not only what your organization does, but why it does it. The “how it does it” will come later with the various policies & procedures, but this first standard talks about something more basic and philosophical.
Topics: HME Accreditation Requirements